New Material Offers Promise for Solid-State Batteries
A new chlorine-based solid-state electrolyte for lithium-ion batteries could lead to higher safety and increased energy density.
Researchers from the University of Waterloo, Canada, who are members of the Joint Center for Energy Storage Research (JCESR), headquartered at the U.S. Department of Energy's (DOE) Argonne National Laboratory, have discovered a new solid electrolyte that offers several important advantages.
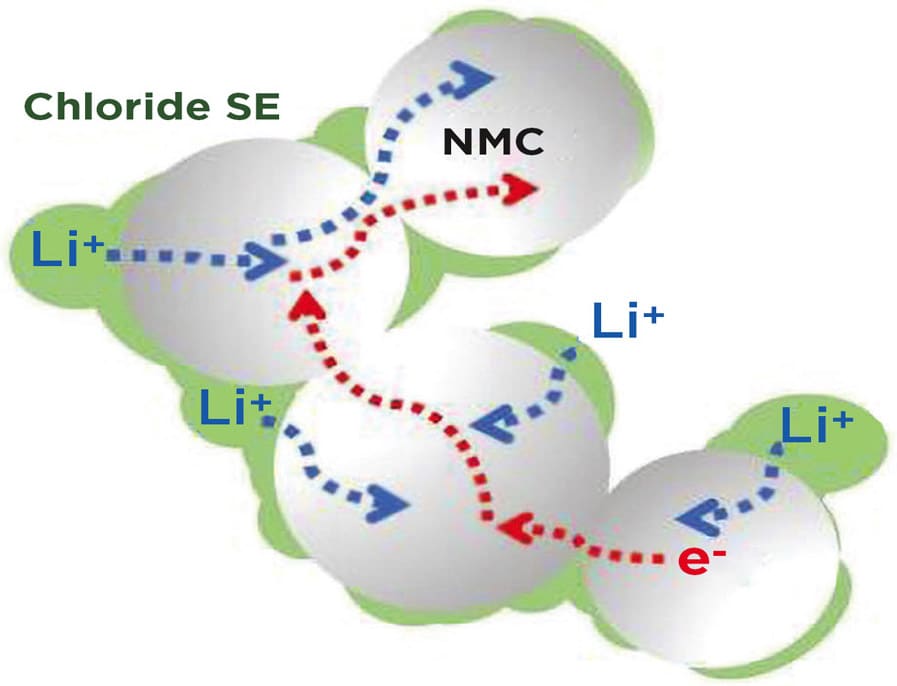
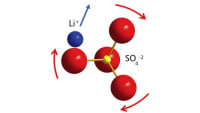
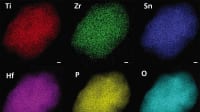
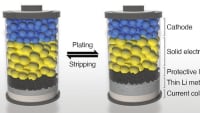
This electrolyte, composed of lithium, scandium, indium, and chlorine, conducts lithium ions well but electrons poorly. This combination is essential to creating an all-solid-state battery that functions without significantly losing capacity for over a hundred cycles at high voltage (above 4 volts) and thousands of cycles at intermediate voltage. The chloride nature of the electrolyte is key to its stability at operating conditions above 4 volts -meaning it is suitable for typical cathode materials that form the mainstay of today's lithium-ion cells.
Current iterations of solid-state electrolytes focus heavily on sulfides, which oxidize and degrade above 2.5 volts. Therefore, they require the incorporation of an insulating coating around the cathode material that operates above 4 volts, which impairs the ability of electrons and lithium ions to move from the electrolyte and into the cathode.
The team wasn't the first to devise a chloride electrolyte, the decision to swap out half of the indium for scandium based on their previous work proved to be a winner in terms of lower electronic and higher ionic conductivity.
One chemical key to the ionic conductivity lay in the material's crisscrossing 3D structure called a spinel. The researchers had to balance two competing desires – to load the spinel with as many charge carrying ions as possible, but also to leave sites open for the ions to move through.
It is not yet clear why the electronic conductivity is lower than many previously reported chloride electrolytes, but it helps establish a clean interface between the cathode material and solid electrolyte, a fact that is largely responsible for the stable performance even with high amounts of active material in the cathode.
The research was funded by the DOE's Office of Science and Office of Basic Energy Sciences with some support from Canada's National Sciences and Engineering Research Council.
For more information, contact media@ anl.gov; 630-252-5580.
Top Stories
INSIDERDefense
![]() New Raytheon and Lockheed Martin Agreements Expand Missile Defense Production
New Raytheon and Lockheed Martin Agreements Expand Missile Defense Production
NewsAutomotive
![]() Ford Announces 48-Volt Architecture for Future Electric Truck
Ford Announces 48-Volt Architecture for Future Electric Truck
INSIDERManufacturing & Prototyping
![]() Active Strake System Cuts Cruise Drag, Boosts Flight Efficiency
Active Strake System Cuts Cruise Drag, Boosts Flight Efficiency
ArticlesTransportation
![]() Accelerating Down the Road to Autonomy
Accelerating Down the Road to Autonomy
INSIDERMaterials
![]() How Airbus is Using w-DED to 3D Print Larger Titanium Airplane Parts
How Airbus is Using w-DED to 3D Print Larger Titanium Airplane Parts
Road ReadyTransportation
Webcasts
Electronics & Computers
![]() Cooling a New Generation of Aerospace and Defense Embedded...
Cooling a New Generation of Aerospace and Defense Embedded...
Power
![]() Battery Abuse Testing: Pushing to Failure
Battery Abuse Testing: Pushing to Failure
Connectivity
![]() A FREE Two-Day Event Dedicated to Connected Mobility
A FREE Two-Day Event Dedicated to Connected Mobility
Automotive
![]() Quiet, Please: NVH Improvement Opportunities in the Early Design Cycle
Quiet, Please: NVH Improvement Opportunities in the Early Design Cycle
Transportation
![]() Advantages of Smart Power Distribution Unit Design for Automotive &...
Advantages of Smart Power Distribution Unit Design for Automotive &...
Aerospace
![]() Sesame Solar's Nanogrid Tech Promises Major Gains in Drone Endurance
Sesame Solar's Nanogrid Tech Promises Major Gains in Drone Endurance