Eco-Friendly Upcycling: Turning Spent Batteries into High-Voltage Energy Storage Systems
A new electrochemical upcycling process enables direct use of LiMn2O4 from spent lithium-ion batteries in zinc-manganese redox flow batteries, significantly reducing energy consumption and environmental impact compared to conventional recycling.
As electric vehicles and energy storage systems (ESS) become increasingly widespread, the management and recycling of spent lithium-ion batteries has emerged as a pressing global issue. Traditional recycling methods, such as energy-intensive smelting or chemically aggressive wet processes, require significant energy and pose environmental risks.
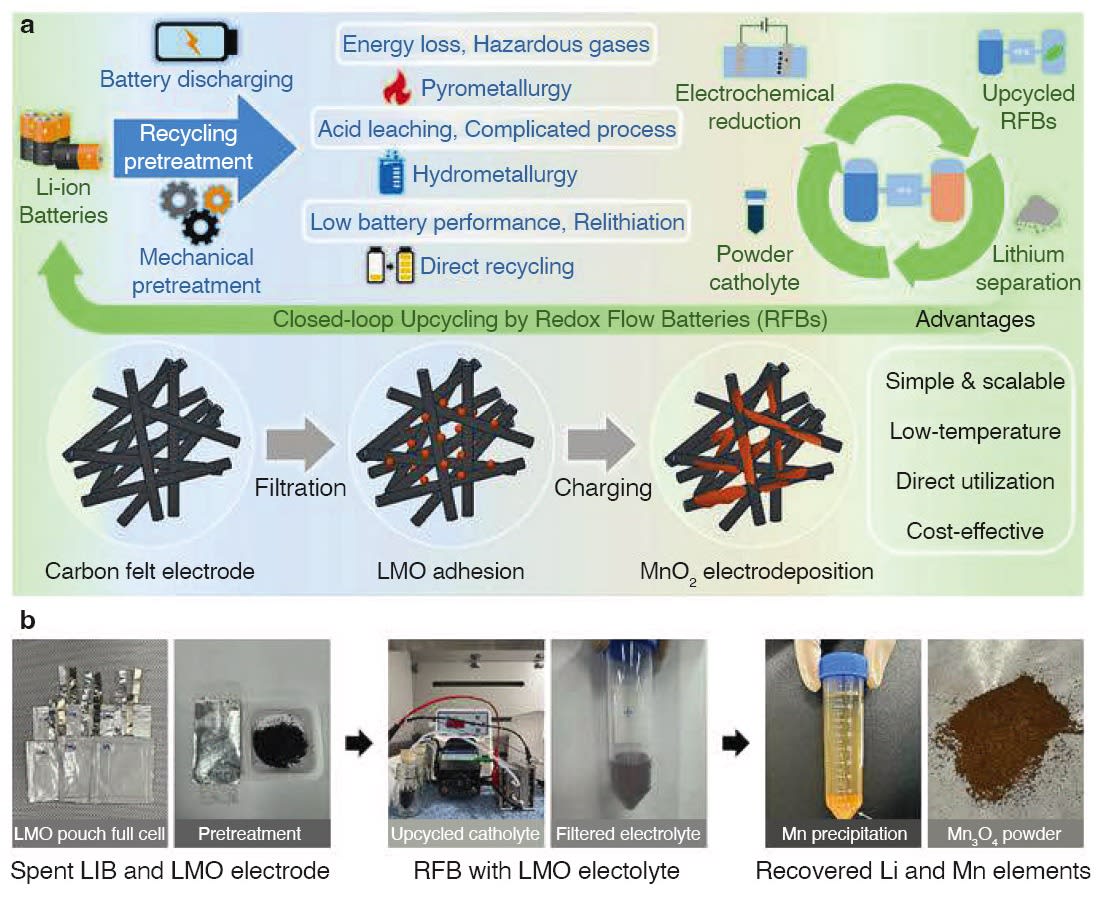
A research team led by Dr. Yosep Han at the Korea Institute of Geoscience and Mineral Resources (KIGAM) has successfully developed an eco-friendly electrochemical process to upcycle lithium manganese oxide (LiMn2O4, LMO), a common cathode material in spent lithium-ion batteries. This process was directly integrated into a zinc–manganese redox flow battery (Zn–Mn RFB), a promising next-generation energy storage system, demonstrating its practical feasibility.
Unlike conventional recycling that focuses on metal recovery, this method electrochemically converts LMO into manganese ions (Mn2+), which are then used as electrolytes for redox flow batteries. The team’s innovation represents a substantial shift toward value-added recycling, moving beyond simple resource recovery, enabling a circular battery ecosystem.
This approach also allows manganese and lithium to be selectively separated by simply adjusting the electrolyte’s pH, further facilitating material reuse. The technology enables spent batteries to serve as a direct source of electrolyte and subsequently be reconverted into precursor materials for new batteries — laying the groundwork for a sustainable, closed-loop battery lifecycle.
Traditional recovery processes typically rely on high-temperature (over 900 °C) smelting or strong-acid-based hydrometallurgy, which require substantial energy and pose environmental risks. In contrast, the new method developed by KIGAM eliminates the need for thermal or chemical extremes, significantly reducing both energy consumption and ecological impact.
Rather than decomposing the LMO material, the researchers guided it through an electrochemical conversion into Mn2+ ions and integrated it into the battery’s electrolyte. The result: comparable initial performance to commercial MnSO4-based electrolytes, and over 70 percent energy efficiency retained after 250 charge/discharge cycles.
Moreover, the team applied a dual-membrane hybrid redox flow battery architecture to achieve high operating voltage and extended cycle life—key requirements for the commercialization of large-scale, long-duration energy storage systems.
“This research overcomes the complexity and environmental drawbacks of existing battery recycling technologies,” said Dr. Yosep Han. “We aim to further enhance battery resource circularity and energy storage efficiency, contributing to carbon neutrality and a recycling-oriented society.”
For more information, contact Yosep Han at
Top Stories
NewsRF & Microwave Electronics
![]() Microvision Aquires Luminar, Plans Relationship Restoration, Multi-industry Push
Microvision Aquires Luminar, Plans Relationship Restoration, Multi-industry Push
INSIDERAerospace
![]() A Next Generation Helmet System for Navy Pilots
A Next Generation Helmet System for Navy Pilots
INSIDERDesign
![]() New Raytheon and Lockheed Martin Agreements Expand Missile Defense Production
New Raytheon and Lockheed Martin Agreements Expand Missile Defense Production
INSIDERMaterials
![]() How Airbus is Using w-DED to 3D Print Larger Titanium Airplane Parts
How Airbus is Using w-DED to 3D Print Larger Titanium Airplane Parts
NewsPower
![]() Ford Announces 48-Volt Architecture for Future Electric Truck
Ford Announces 48-Volt Architecture for Future Electric Truck
ArticlesAR/AI
Webcasts
Electronics & Computers
![]() Cooling a New Generation of Aerospace and Defense Embedded...
Cooling a New Generation of Aerospace and Defense Embedded...
Automotive
![]() Battery Abuse Testing: Pushing to Failure
Battery Abuse Testing: Pushing to Failure
Power
![]() A FREE Two-Day Event Dedicated to Connected Mobility
A FREE Two-Day Event Dedicated to Connected Mobility
Unmanned Systems
![]() Quiet, Please: NVH Improvement Opportunities in the Early Design Cycle
Quiet, Please: NVH Improvement Opportunities in the Early Design Cycle
Automotive
![]() Advantages of Smart Power Distribution Unit Design for Automotive &...
Advantages of Smart Power Distribution Unit Design for Automotive &...
Energy
![]() Sesame Solar's Nanogrid Tech Promises Major Gains in Drone Endurance
Sesame Solar's Nanogrid Tech Promises Major Gains in Drone Endurance